Previously, we discussed how proteomics is a better measure of the pharmacodynamic effects of target inhibition in drug development. There are a few ways to analyze protein levels and function across patients samples including Mass Spectrometry, ELISA, and our specialty, Reverse Phase Protein Array (RPPA). Today we’ll be giving an overview and comparison of a few of these methods:
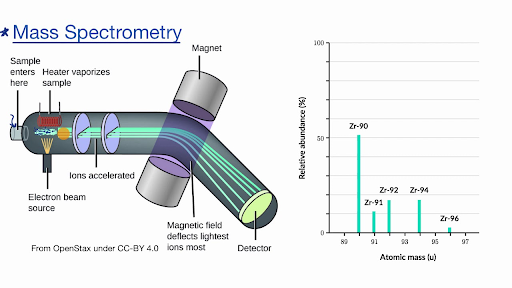
Mass Spectrometry
Mass Spectrometry gives a look at the entire proteomic landscape of a sample. It is great for the identification and quantification of wild-type and mutant proteins. It also helps to identify post-translational modifications and the presence of splice variants. However, oftentimes mass spectrometry is not sensitive enough to detect low-level signals. A recent analysis of human ovarian and breast cancer xenograft tissue detected only about 60% of identified phosphorylation, of which even fewer could be quantified by mass spectrometry. The use of mass spectrometry requires quite a lot of sample material in addition to expensive equipment and highly trained operators. This limits analysis to a few specialized centers that offer services to many CLIA laboratories.
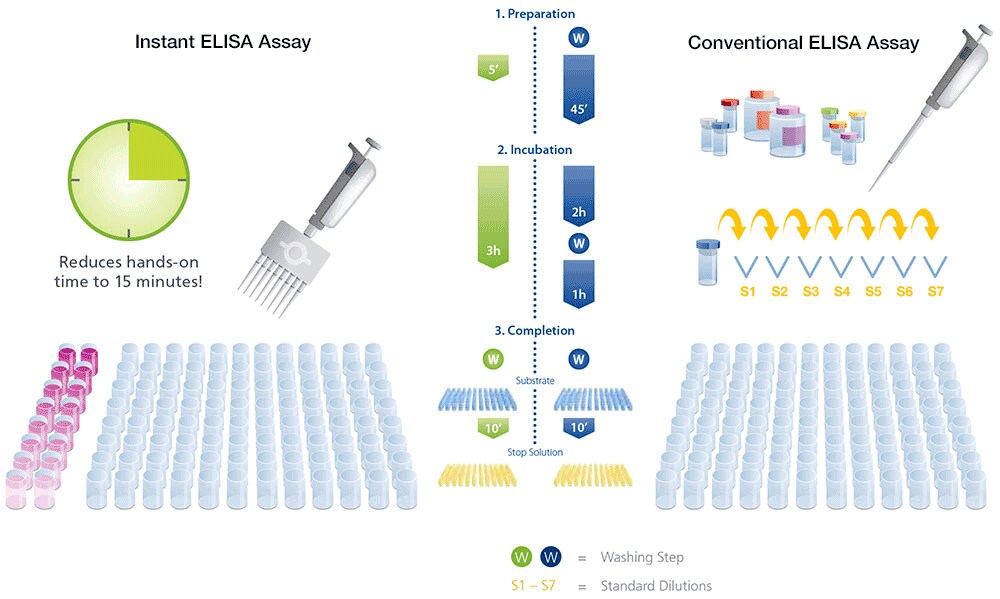
Enzyme-Linked Immunosorbent Assay (ELISA)
An ELISA is a plate-based assay that can detect and quantify soluble native proteins, and protein interactions that require intact three-dimensional structure. The assay is not highly specific and sometimes produces false positives due to the cross-reactivity of the detecting antibody with other proteins in the sample. ELISAs can be adapted to higher throughput with automated plate handling and detection systems.
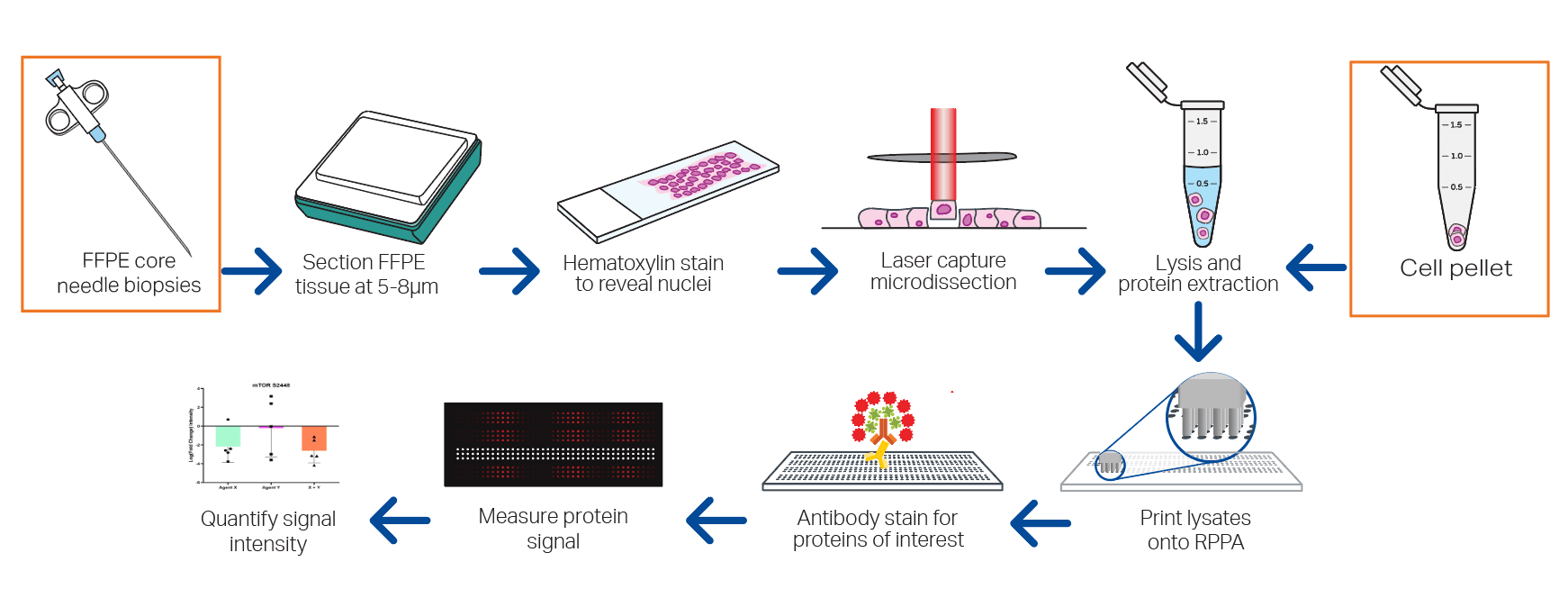
Reverse Phase Protein Array (RPPA)
RPPA is just like a dot blot/ western blot except you blot the sample down a slide and not a gel. Unlike a dot blot, is less labor-intensive, much more high-throughput, very sensitive. At Theralink, we have over 700 validated protein and phosphoprotein antibodies, 100% customizable pathways for your mechanism of action (MOA), quantitative adn senstivie multiplex capacity, and translatable and predictive data.
RPPA Vs Mass Spectrometry
- For discovery ventures where one is unsure of the target of interest and is checking for the presence or absence of protein, it is best to use Mass Spectrometry, however, with a known target RPPA offers a more cost-effective and robust way of assessing the activated proteome.
- If you have a high abundance target then Mass Spectrometry is great, however for post-translational modifications where the protein isn’t abundant then Mass Spectrometry does not meet the required sensitivity. RPPA often provides better sensitivity at lower concentrations.
RPPA Vs ELISA
- Our Theralink platform uses denatured protein and not native proteins like ELISA does.
- While we have the ability to work with FFPE samples, ELISAs cannot.
- ELISAs are good for looking for proteins in solution vs RPPA which is best for looking at protein from tissue extracts which is often the case for patient samples.
- With an ELISA you often need both primary and secondary antibodies so you are limited by market availability vs RPPA that works with any antibody designed for western blot.
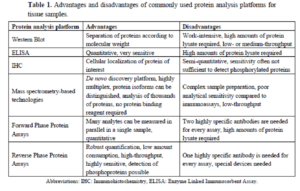
RPPA Vs Other Protein Analysis Platforms
There are other commonly used protein analysis platforms and in this review by Stephanie Boellner and Karl-Friedrick Becker, the various platforms were tabularly compared: