BioMed Valley Discoveries recently shared their latest findings at AACR 2022 regarding combining ulixertinib (ERK1/2 inhibitor) and CDK4/6 inhibitors to treat MAPK pathway altered tumors. As part of this work, Theralink’s RPPA technology was used to understand how administration of the first-in-class small molecule inhibitor of ERK1/2, ulixertinib (BVD-523), combined with the CDK4/6 inhibitor palbociclib, impacted cell signaling pathways to inhibit tumor growth. Here, we’ll take a closer look at the findings and reveal how we were able to contribute.
The MAPK/ERK Pathway
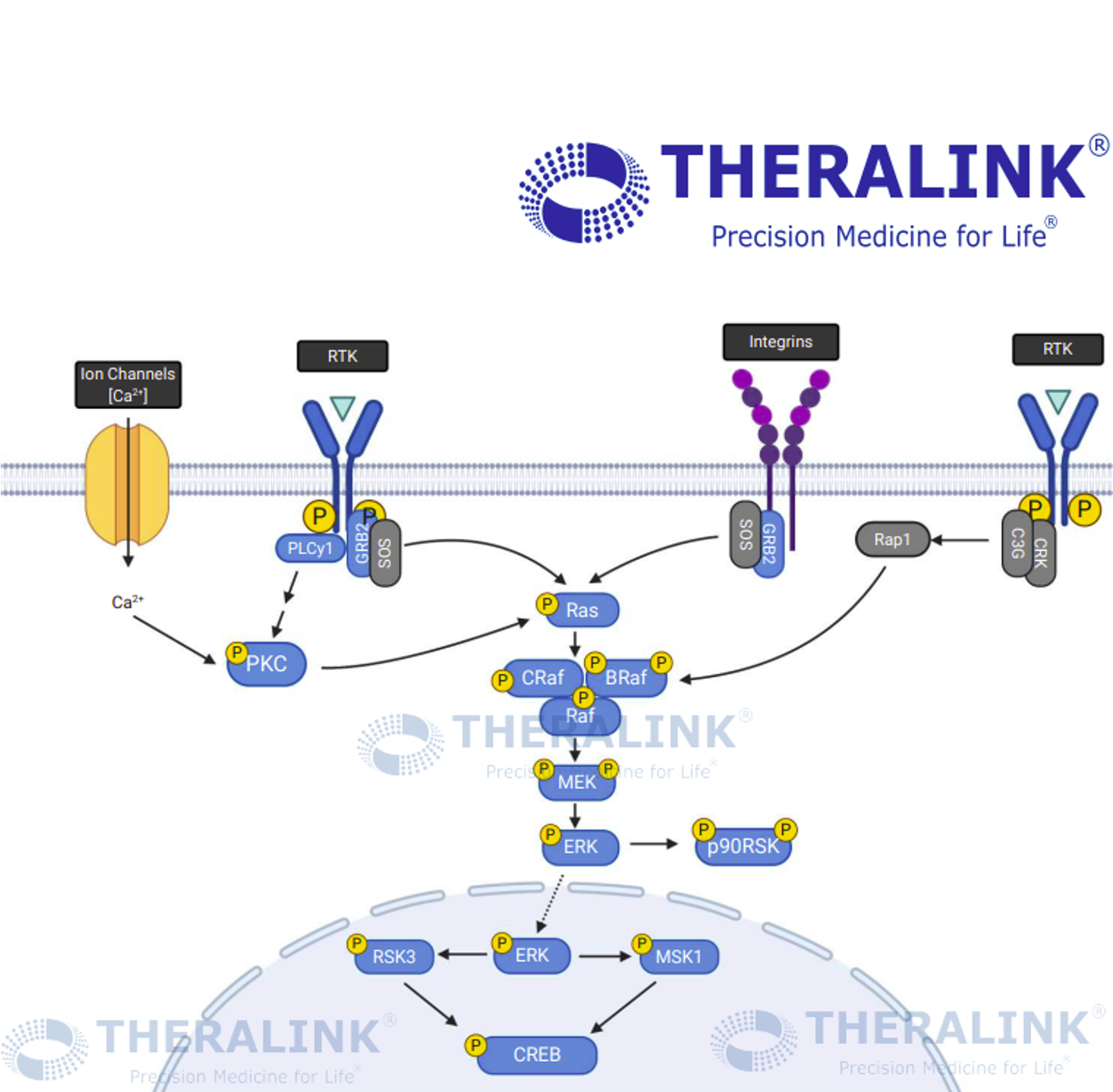
First, let’s give a brief overview of the MAPK/ERK pathway (Figure 1). In the cell, this pathway regulates cell growth and proliferation through a phosphorylation cascade system. Activated membrane-bound receptor tyrosine kinases activate RAS proteins, which in turn activate the ERK protein s via RAF/MEK. ERK activation induces cell proliferation and survival through multiple mechanisms, including increasing cyclin D1 levels and entry into the cell cycle, in addition to activation of numerous transcription factors like c-Myc and ELK. When the MAPK/ERK pathway is activated and cell cycle is dysregulated, cancer can result.
To combat this, multiple drugs have been developed targeting various points in the pathway. Ulixertinib (also known as BVD-523) does so by inhibiting ERK activity, while palbociclib prevents cell cycle progression by inhibiting CDK4/6 kinases. Researchers at BioMed Valley Discoveries decided to investigate whether combining palbociclib and ulixertinib might improve outcomes over single-drug treatments, using Theralink’s Reverse-Phase Protein Array (RPPA) platform to investigate the cell signaling effects.
Combination treatment significantly inhibited tumor growth
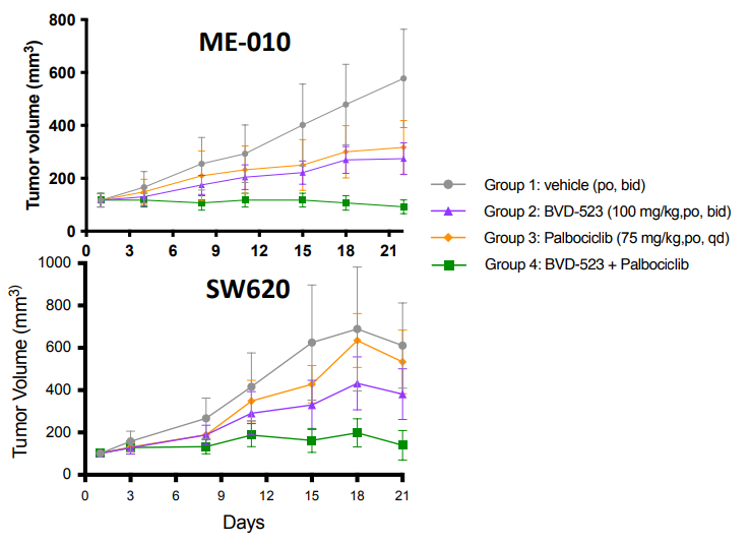
Using cell-line derived xenographs (CDX ) and patient derived xenographs (PDX), researchers found that while palbociclib or ulixertinib alone slowed the growth of the tumor when compared to a vehicle control, the combination treatment demonstrated significant responses ranging from 75%-90% tumor growth inhibition after 21 days (Figure 2). These results, along with other supporting evidence, suggests that the combination treatment is highly effective at preventing tumor growth. But what was happening at the cellular level?
RPPA results show combination therapy inhibits cellular signaling pathways distinctly
To understand how these treatments impacted cell signaling, we used our RPPA platform to analyze CDX that were flash-frozen (FF). We then used laser capture microdissection to isolate tumor epithelium and used a custom panel of 24 analytes to examine the MAPK/ERK pathway, potential resistance mechanisms, and downstream cell proliferation targets. In addition to total analyte levels, we measured phosphorylation status of multiple analytes to measure the activation levels of these pathways.
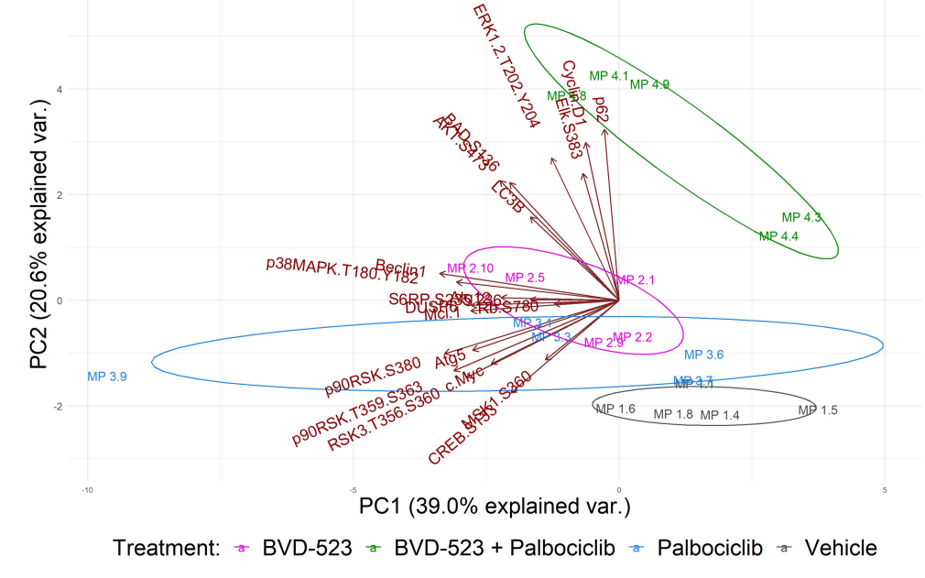
Using Principal Component Analysis (PCA), we see that samples within a treatment group cluster together, indicating that samples within a treatment group produce similar results (Figure 3). Furthermore, the combination therapy treatment clusters away from all other treatment group, indicating a distinct treatment response compared to either monotherapy or vehicle control. The multiplexed capabilities of the RPPA platform helped elucidate these differences amongst treatment groups.
RPPA reveals insights on the ERK pathway, downstream transcription factors, and compensatory pathways
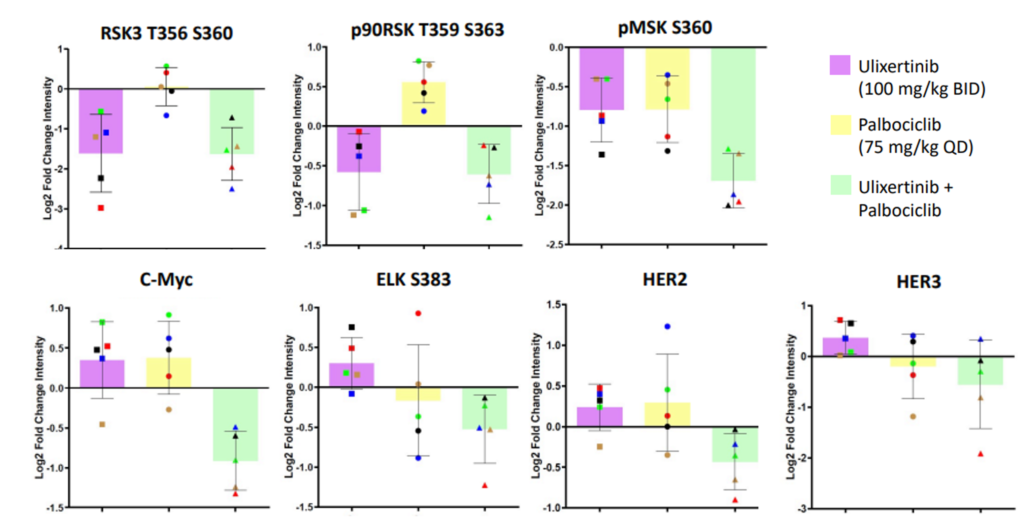
While PCA analysis can give us broad insights into what is happening inside the cell, we can also visualize RPPA data at the individual protein level (Figure 4). Examining the downstream targets of ERK, both single agent ulixertinib and combination therapy reduce phosphorylation levels of p90RSK T359 and S363; as well as RSK3 T356 and S360. This reduction is not seen when single agent palbociclib is administered, indicating that ulixertinib is the driving force behind these trends. However, the combined therapy deceased MSK S360 phosphorylation more than either single agent, suggesting a combination effect.
When we look at C-Myc and ELK (two downstream cell proliferation transcription factors), protein levels are reduced in the combination therapy compared to either monotherapy, suggesting a strong combination effect as well. Finally, we looked at the alternate cell proliferation pathway, HER2/HER3. Under MAPK pathway inhibition, HER proteins can sometimes become activated to act as an escape mechanism to allow continued cell proliferation. However, under the combination treatment we see no dramatic increase in HER2 or HER3, suggesting the combination treatment is resistant to this escape mechanism.
The Theralink RPPA Platform
Whether you are in the earliest discovery stages or performing clinical trials, let Theralink expedite your discoveries with in-depth knowledge of metabolic responses and compensatory pathways. With over 700 analytes across dozens of pathways already validated and the ability to add your custom biomarker panels, we can ensure your specific project needs are met.